Health
Over Three Dozen Babies Affected by Botulism Linked to Formula

More than three dozen infants have been affected by an outbreak of infant botulism linked to contaminated formula produced by ByHeart, a New York-based manufacturer. The Food and Drug Administration (FDA) reported on November 8, 2023, that there have been 37 confirmed or suspected cases of illness in babies who consumed ByHeart formula since August. The most recent illness was reported on November 19, with all affected infants requiring hospitalization. Fortunately, no fatalities have been documented in this outbreak.
Following the FDA’s findings, ByHeart issued a recall of all its products sold in the United States on November 11. The company, which holds approximately 1% of the U.S. infant formula market, was distributing about 200,000 cans of its product monthly. FDA inspectors have conducted visits to the company’s production facilities located in Allerton, Iowa, and Portland, Oregon, to investigate the circumstances surrounding the contamination.
The outbreak primarily involves infants aged between 2 weeks and 9 months. According to the FDA, the illnesses began between August 9 and November 19, affecting babies across multiple states, including Arizona, California, and Texas. A sample taken from an open can of ByHeart formula in California contained the bacteria responsible for infant botulism, reinforcing the link between the product and the reported cases.
The FDA is currently investigating a notable increase in infant botulism cases since August. The agency stated, “ByHeart brand formula is disproportionately represented among sick infants in this outbreak.” Medical experts emphasize that symptoms of this condition can take up to 30 days to manifest and may include poor feeding, loss of head control, drooping eyelids, and a flat facial expression. Infants can become “floppy” and experience difficulties with swallowing or breathing.
In response to the incidents, families of affected infants have filed lawsuits against ByHeart, alleging that the formula provided was defective and that the company acted negligently in its sale. The lawsuits, lodged in federal courts, seek financial compensation for medical expenses, emotional distress, and other damages resulting from the illnesses.
Typically, fewer than 200 cases of infant botulism are reported annually in the United States. The Centers for Disease Control and Prevention (CDC) indicated that 133 cases had been documented as of September 20, 2023. This compares to 145 cases reported throughout the entirety of the previous year. The condition arises from a type of bacteria that produces toxins in the large intestine, with infants being particularly vulnerable due to their immature gut microbiomes. They can contract the infection through exposure to spores found in dirt, dust, or contaminated honey.
Historically, there have been sporadic links between baby formula and illness, but this incident marks the first confirmed outbreak of infant botulism associated with powdered formula. The only known treatment for the condition is BabyBIG, an intravenous medication derived from the blood plasma of adults who have been immunized against botulism. Developed by California’s infant botulism program, BabyBIG is the sole source of this treatment worldwide and aims to reduce hospital stays and the severity of the illness.
Concerns about infant formula shortages are minimal, given that ByHeart represents a small segment of the market. This stands in stark contrast to the crises experienced in late 2021 and 2022, when a different germ caused severe illness in four infants, resulting in two deaths. That situation prompted the FDA to shut down Abbott Nutrition’s Michigan plant after contamination issues were identified.
In 2022, ByHeart had previously recalled five batches of infant formula after a sample tested positive for cronobacter sakazakii, another harmful bacterium. In 2023, the FDA issued a warning letter to ByHeart detailing necessary corrective actions, following the discovery of mold, water leaks, and insects during an inspection at a Pennsylvania plant.
In light of recent events, federal health officials are committed to reforming the U.S. food supply. Health Secretary Robert F. Kennedy Jr. has directed the FDA to undertake a comprehensive review of the nutrients and ingredients present in infant formula. This initiative, referred to as “Operation Stork Speed,” marks the first significant examination of formula ingredients in over 25 years. The FDA is currently reviewing feedback from industry stakeholders, health experts, and the public to determine the next steps in ensuring the safety of infant nutrition.
-

 Science4 weeks ago
Science4 weeks agoUniversity of Hawaiʻi at Mānoa Joins $25.6M AI Initiative for Disaster Monitoring
-

 Science2 months ago
Science2 months agoIROS 2025 to Showcase Cutting-Edge Robotics Innovations in China
-
 Science2 weeks ago
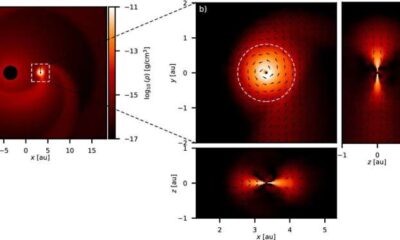
Science2 weeks agoALMA Discovers Companion Orbiting Red Giant Star π 1 Gruis
-

 Lifestyle2 months ago
Lifestyle2 months agoStone Island’s Logo Worn by Extremists Sparks Brand Dilemma
-

 Health2 months ago
Health2 months agoStartup Liberate Bio Secures $31 Million for Next-Gen Therapies
-

 Lifestyle2 months ago
Lifestyle2 months agoMary Morgan Jackson Crowned Little Miss National Peanut Festival 2025
-

 World2 months ago
World2 months agoBravo Company Veterans Honored with Bronze Medals After 56 Years
-

 Politics2 months ago
Politics2 months agoJudge Considers Dismissal of Chelsea Housing Case Citing AI Flaws
-

 Health2 months ago
Health2 months agoTop Hyaluronic Acid Serums for Radiant Skin in 2025
-

 Science2 months ago
Science2 months agoArizona State University Transforms Programming Education Approach
-

 Sports2 months ago
Sports2 months agoYamamoto’s Mastery Leads Dodgers to 5-1 Victory in NLCS Game 2
-

 Sports2 months ago
Sports2 months agoMel Kiper Jr. Reveals Top 25 Prospects for 2026 NFL Draft