World
Infant Botulism Cases Linked to ByHeart Formula Reach 31

The outbreak of infant botulism linked to ByHeart infant formula has escalated, with the number of reported cases now at 31 across 15 states. According to the U.S. Food and Drug Administration (FDA), eight additional infants have been hospitalized due to the potentially life-threatening condition, prompting urgent warnings about the recalled formula still being found on some retail shelves.
The FDA’s investigation into this serious health risk has revealed that the affected infants exhibited symptoms consistent with botulism, a rare but serious illness caused by a toxin produced by the bacterium *Clostridium botulinum*. This toxin affects the nervous system and can lead to severe complications, including paralysis. The agency has urged parents and caregivers to be vigilant and check their inventory for the recalled ByHeart products.
Ongoing Risks and Warnings
Despite the recall, the FDA has expressed concern that some stores may still have the affected formula available for purchase. The agency is working closely with ByHeart to ensure that all recalled products are removed from the market. Parents are advised to refrain from using any ByHeart formula manufactured prior to September 2023, as this is when the potential contamination was identified.
The FDA noted that infant botulism typically presents with symptoms such as weakness, poor feeding, constipation, and respiratory distress. Health professionals emphasize that early recognition and treatment are crucial in managing the condition effectively.
In light of this outbreak, the FDA has reiterated the importance of using safe feeding practices and ensuring that any formula used is from reliable sources. Parents are encouraged to consult with their pediatricians regarding any concerns about infant nutrition and safety.
Public Health Implications
This incident underscores the ongoing challenges faced in ensuring the safety of infant feeding products. The FDA’s actions reflect a commitment to public health and safety, as they continue to monitor the situation and work to prevent further cases of botulism linked to formula products.
As investigations continue, health officials stress the need for transparency and swift action when it comes to product recalls. The ByHeart case serves as a reminder of the critical nature of food safety, particularly for vulnerable populations such as infants.
In the coming weeks, the FDA intends to provide further updates on the situation as more information becomes available. Parents and caregivers are urged to stay informed and prioritize their children’s health during this critical time.
-

 Science4 weeks ago
Science4 weeks agoUniversity of Hawaiʻi at Mānoa Joins $25.6M AI Initiative for Disaster Monitoring
-

 Science2 months ago
Science2 months agoIROS 2025 to Showcase Cutting-Edge Robotics Innovations in China
-
 Science2 weeks ago
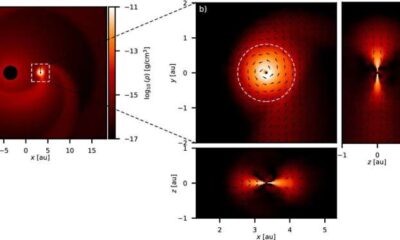
Science2 weeks agoALMA Discovers Companion Orbiting Red Giant Star π 1 Gruis
-

 Lifestyle2 months ago
Lifestyle2 months agoStone Island’s Logo Worn by Extremists Sparks Brand Dilemma
-

 Health2 months ago
Health2 months agoStartup Liberate Bio Secures $31 Million for Next-Gen Therapies
-

 Lifestyle2 months ago
Lifestyle2 months agoMary Morgan Jackson Crowned Little Miss National Peanut Festival 2025
-

 World2 months ago
World2 months agoBravo Company Veterans Honored with Bronze Medals After 56 Years
-

 Politics2 months ago
Politics2 months agoJudge Considers Dismissal of Chelsea Housing Case Citing AI Flaws
-

 Health2 months ago
Health2 months agoTop Hyaluronic Acid Serums for Radiant Skin in 2025
-

 Sports2 months ago
Sports2 months agoYamamoto’s Mastery Leads Dodgers to 5-1 Victory in NLCS Game 2
-

 Science2 months ago
Science2 months agoArizona State University Transforms Programming Education Approach
-

 Business2 months ago
Business2 months agoTruist Financial Increases Stake in Global X Variable Rate ETF